The True Flow Calibration of Ultrasound Diagnostic Instruments
Doppler Ultrasound
by | AAMI 1990 | Publications, Doppler Ultrasound
The True Flow Calibration of Ultrasound Diagnostic Instruments
25th Annual Meeting of the Association for the Advancement of Medical Instrumentation, Proceedings, 22, (1990)
J. C. Conti, Ph.D., E. R. Strope, Ph.D., D. J. Rohde
Dynatek Laboratories, Inc., Galena, MO
Presentation Slides
Association for the Advancement of Medical Instrumentation, 1990
Presented by James C. Conti
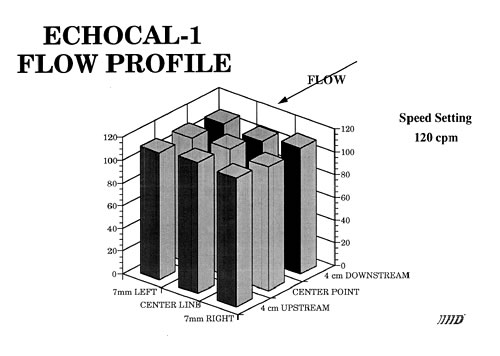
EchoCal
We are entering an exciting period for the field of noninvasive cardiovascular diagnosis. Several state of the art ultrasound instruments are capable of evaluating a variety of cardiac performance characteristics. In addition the FDA is considering heart valve submissions with ultrasound flow analysis instead of indwelling catheter data.
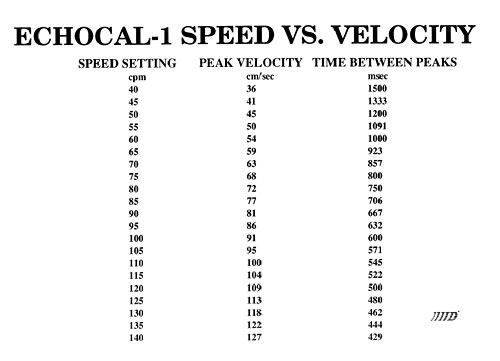
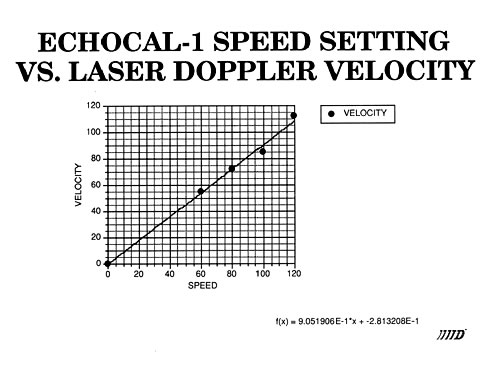
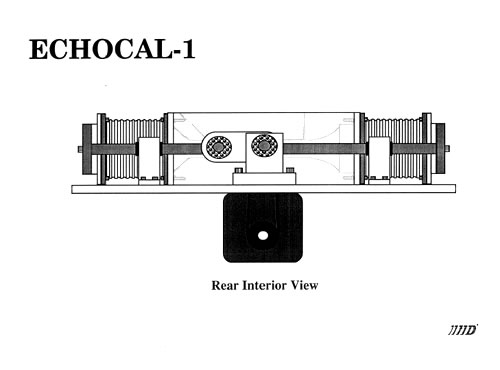
An instrument has been designed and fabricated that develops well-defined and reproducible flow characteristics similar to those found in the human aorta. Sensitivity to cardiac output, flow and natural geometry allow a user to calibrate an ultrasound instrument in an absolute manner.
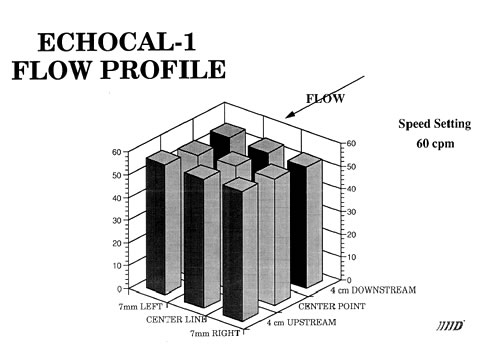
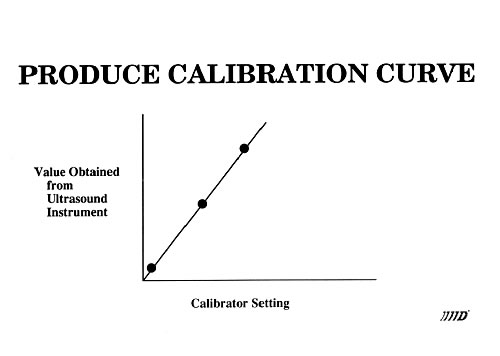
Catheter tipped blood flowmeter data will be presented that demonstrates the reliability and reproducibility of this instrument. An actual flow calibration will then be outlined. In addition, design considerations will be reviewed. These include the requirement that flow be laminar through the measurement region. This is accomplished by use of both a flow nozzling technique and stainless steel screen flow straighteners.
Details of the considerations that must be addressed in the design of such an instrument will be presented.
Dynatek Laboratories, Inc., Fourth and Main, PO Box 254, Galena, MO 65656